No molecular orbital diagram9/9/2023  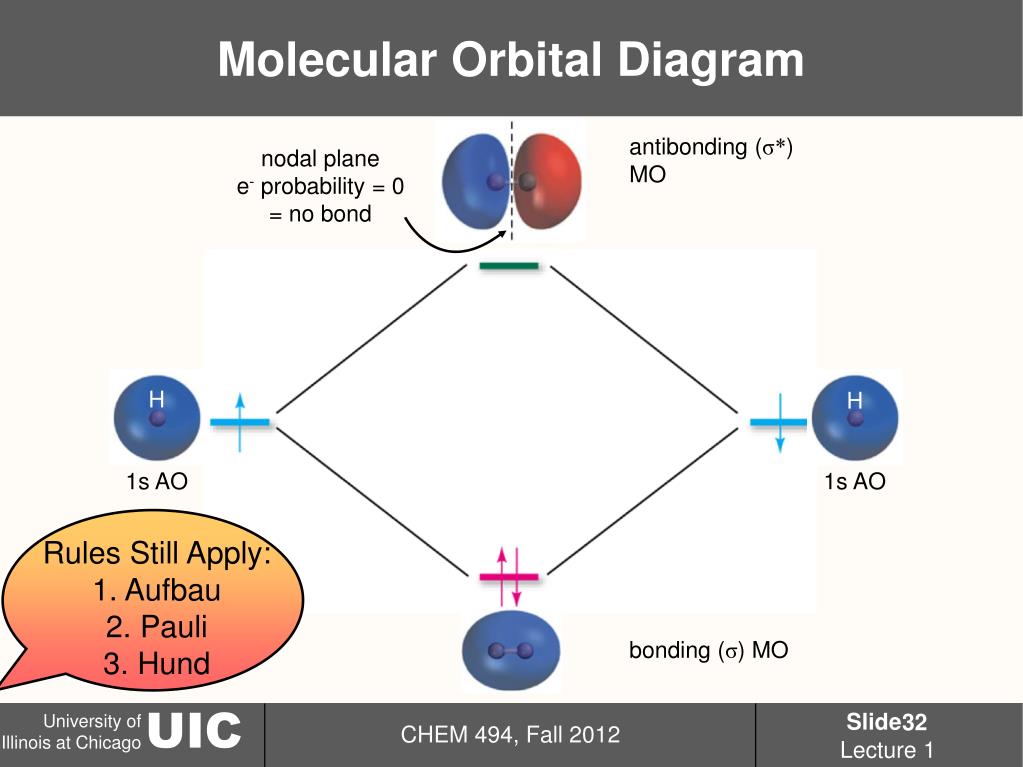
Similar to atomic orbitals, we can write electron configuration energy diagrams for molecular orbitals (Figure 9.20 “Hydrogen molecular orbital electron configuration energy diagram”). Figure 9.19 “Hydrogen molecular orbital combination diagram.” This node of zero electron density is destabilizing toward the bond, making it higher energy, and subsequently this type of orbital is known as an antibonding molecular orbital (denoted by the asterisk in the orbital name). This type of combination results in an area of zero electron density between the two nuclei, known as a nodal plane (or node). The σ* 1 s orbital is generated by a destructive combination (or interference), where the wave functions of the two atomic orbitals cancel each other. These electrons are stabilized by attractions to both nuclei, and they hold the atoms together with a covalent bond. Notice in Figure 9.19 “Hydrogen molecular orbital combination diagram” that the electron density of this orbital is concentrated between the two nuclei. This is the lower energy of the two molecular orbitals and is known as the bonding molecular orbital. The σ 1 s orbital is generated by a constructive combination (or interference), where the two atomic orbitals wave functions reinforce (add to) each other. The Molecular Orbitals of the Hydrogen MoleculeĬombining the 1 s orbitals of each hydrogen atom using LCAO, two molecular orbitals are generated σ 1 s (pronounced sigma one s) and σ* 1 s (pronounced sigma star one s). 
Molecular orbitals are associated with the entire molecule, meaning the electron density is delocalized (spread out) over more than one atom. The major difference between atomic and molecular orbitals is that atomic orbitals represent electron density in space associated with a particular atom.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
